Filana Therapeutics Announces Publication in Epilepsia of Preclinical Simufilam Data
- Study in a well-accepted mouse model of severe Tuberous Sclerosis Complex (TSC) showed that simufilam attenuated the progression of seizures in a dose-dependent manner
- Results confirm and extend previously published preclinical findings
- TSC-related epilepsy affects approximately 45,000 people in the U.S.
AUSTIN, Texas, April 15, 2026 (GLOBE NEWSWIRE) -- Filana Therapeutics, Inc. (NASDAQ: FLNA, “Filana Therapeutics”, the “Company”), a biotechnology company focused on developing therapies for Tuberous Sclerosis Complex (TSC)-related epilepsy, today announced publication in Epilepsia of preclinical data showing that simufilam attenuated seizure progression in a well-accepted mouse model of severe TSC-related epilepsy1. The results support the continued evaluation of simufilam for the treatment of TSC-related epilepsy, which affects approximately 45,000 people in the U.S.2,3. Simufilam is an oral small molecule intended to modulate filamin A protein.
The study was led by Angélique Bordey, PhD, Senior Vice President, Neuroscience of Filana Therapeutics and Vice Chair of Research for the Department of Neurosurgery at Yale School of Medicine, and included a co‑author from Yale School of Medicine’s Departments of Neurosurgery and Cellular & Molecular Physiology. Top line results of the study were previously reported by the Company in August 2025. The work was conducted in collaboration with the TSC Alliance® and PsychoGenics, Inc., the clinical research partner of the TSC Preclinical Consortium. The paper, titled “The small molecule simufilam dose-dependently attenuates the worsening of seizures in a mouse model of tuberous sclerosis complex1” was published online in Epilepsia on April 14, 2026, and is available at https://doi.org/10.1002/epi.70227.
This dose escalation study evaluated simufilam’s ability to slow the worsening of seizures in the Tsc1 conditional knockout mouse (Tsc1-cKO)3, an exceptionally severe and progressive model of TSC-related epilepsy. Seizures were monitored for approximately three weeks following onset. The data showed that simufilam attenuated the progression of seizure activity compared to vehicle, with a statistically significant correlation between simufilam dose and the number of seizures by the end of the study.
“The formal publication of these data in Epilepsia supports our previously reported findings and reinforces the biological rationale for studying simufilam in TSC-related epilepsy,” said Dr. Bordey. “Together with our findings in an earlier animal model, these results underscore the positive correlation between seizure outcomes and plasma exposure to simufilam and bolster the scientific basis of our clinical program.”
“While we continue to address FDA’s questions related to the clinical hold, the publication of our data in Epilepsia adds to the biological rationale behind simufilam in TSC-related epilepsy,” said Rick Barry, President and Chief Executive Officer of Filana Therapeutics, Inc. “We remain focused on generating the necessary data to resolve the clinical hold and advancing our program for the TSC community.”
Key Takeaways
The study evaluated simufilam in the Tsc1-cKO mouse, an exceptionally severe and rapidly progressive model of TSC-related epilepsy. In this model, seizures normally worsen steadily over time.
- Simufilam attenuated the expected worsening of seizures over time in both number and total duration
- Attenuation of seizure worsening increased with drug exposure
Figure 3C from the article is a heatmap comparing the number of seizures per day recorded for each mouse in the study. Mice were administered vehicle or simufilam at one of three ascending doses.
Epilepsia paper, Figure 3C
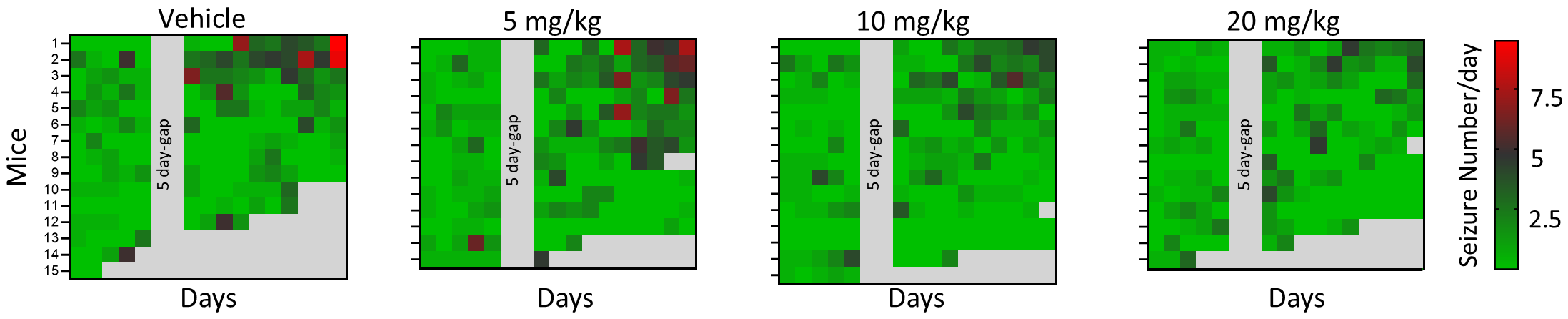
Heatmap of the daily number of seizures per mouse during EEG recordings in each condition. EEG monitoring spanned 20 days, but seizure counts were quantified for the first 5 and last 10 days, and comparative analysis was performed between the first and last 5 days. Gray squares: Mice had died. The 5 day-gap indicates that analysis was not performed for this period.
TSC Program Status
Filana Therapeutics is working to address the previously disclosed request for information from FDA that was contained in a December 2025 Clinical Hold Letter, including the submission of additional pre-clinical data and protocol design modifications. The Company intends to submit a response to FDA following completion of these activities and expects to provide a program update in the coming months.
The TSC Alliance
The TSC Alliance is an internationally recognized nonprofit organization dedicated to improving the lives of individuals affected by Tuberous Sclerosis Complex (TSC)-related epilepsy. It drives research towards a cure, accelerates new treatments, and provides comprehensive support and education, while advocating for those affected by the disease. Learn more at www.tscalliance.org.
TSC Preclinical Consortium
The TSC Alliance Preclinical Consortium is a collaborative partnership launched by the TSC Alliance to unite academic and industry researchers to test potential therapies for Tuberous Sclerosis Complex (TSC)-related epilepsy. It accelerates drug development by evaluating new and repurposed compounds in standardized animal models to move promising potential treatments into clinical trials faster.
One of the models offered by the TSC Preclinical Consortium is the Tsc1-cKO mouse model developed by Michael Wong, MD, PhD, at Washington University School of Medicine and housed at PsychoGenics, Inc. (PsychoGenics) in collaboration with the TSC Alliance. This model was used in the experiments reported by Filana Therapeutics in the Epilepsia paper.
About PsychoGenics, Inc.
PsychoGenics is a preclinical Contract Research Organization that specializes in central nervous system and orphan diseases, with a focus on psychiatric and neurological conditions including seizure disorders, Alzheimer’s disease, Parkinson’s disease, schizophrenia and pain.
About TSC and TSC-related Epilepsy
TSC is a rare genetic disorder resulting from a mutation in the TSC1 or TSC2 gene. This affects the mechanistic target of rapamycin (mTOR) pathway and can cause tumors to grow in multiple organs4. Epilepsy is the most common health issue affecting the TSC community, with 80% to 90% of TSC patients experiencing seizures5. TSC-related epilepsy affects approximately 45,000 people in the U.S.2,3 Most patients start having seizures within their first year of life2. Even with multiple approved treatments, more than 60% of TSC patients remain refractory to antiepileptic therapy6.
About Filana Therapeutics, Inc.
Filana Therapeutics, Inc. (NASDAQ: FLNA), is a biotechnology company focused on developing novel therapies to modulate filamin A for the treatment of central nervous system disorders (CNS), such as tuberous sclerosis complex (TSC)-related epilepsy, and other diseases associated with dysregulation or overexpression of filamin A.
For more information, please visit: https://www.FilanaTx.com
References:
- Stansley B, Islam MM, Aguiar DJ, Fuchs Z, Catron M, Morairty S, et al. The small molecule simufilam dose-dependently attenuates the worsening of seizures in a mouse model of tuberous sclerosis complex. Epilepsia. 2026;00:1–13. https://doi.org/10.1002/epi.70227
- Zhang L, Huang T, Teaw S, Nguyen LH, Hsieh LS, Wong X, Burns LH, Bordey A. Filamin A inhibition reduces seizure activity in a mouse model of focal cortical malformations. Sci. Transl. Med. (2020) 12(531). https://www.science.org/doi/10.1126/scitranslmed.aay0289
- https://www.tscalliance.org/understanding-tsc/what-is-tsc/
- https://www.tscalliance.org/researchers/preclinical-research/
- Crino P, Nathanson K, Petri Henske, E. The Tuberous Sclerosis Complex. N Engl J Med. (2006) 355 (13):1345-56. DOI: 10.1056/NEJMra055323
-
Chu-Shore, C. J., Major, P., Camposano, S., Muzykewicz, D., & Thiele, E. A. (2010). The natural history of epilepsy in tuberous sclerosis complex. Epilepsia. 2010; 51(7): 1236–1241. https://doi.org/10.1111/j.1528-1167.2009.02474.x
For More Information Contact:
Investors
Sandya von der Weid
svonderweid@lifesciadvisors.com
Company
Eric Schoen, Chief Financial Officer
(512) 501-2450
ESchoen@FilanaTx.com
IR@FilanaTx.com
Cautionary Note Regarding Forward-Looking Statements:
This news release contains forward-looking statements that may include but are not limited to statements regarding: our ability to successfully engage with, and satisfactorily respond to, requests for additional information from the U.S. Food and Drug Administration (FDA) concerning the full clinical hold on our investigational new drug application (IND) for simufilam in TSC-related epilepsy and the timing and outcomes of such interactions, the timing and plans to conduct clinical studies with simufilam following approval of our IND, our plans to conduct additional preclinical studies of simufilam relating to seizures in TSC, the potential for simufilam as a treatment for TSC-related epilepsy and other potential indications, and the timing of anticipated milestones. These statements may be identified by words such as “anticipate”, “before”, “believe”, “could”, “expect”, “forecast”, “intend”, “may”, ”pending”, “plan”, “possible”, “potential”, “prepares for”, “will”, and other words and terms of similar meaning.
Such statements are based on our current expectations and projections about future events. Such statements speak only as of the date of this news release and are subject to a number of risks, uncertainties and assumptions, including, but not limited to, those risks relating to our ability to provide FDA with additional information, including additional pre-clinical data, and modifying the protocol design proposed clinical trial, to satisfy completion of FDA’s review and release of full clinical hold, the ability to advance preclinical studies related to TSC-related epilepsy, and other potential indications, the ability to successfully carry out the Company’s obligations under the Yale License Agreement, the ability to initiate an initial proof-of-concept study of simufilam in TSC-related epilepsy, and other risks inherent in drug discovery and development or specific to Filana Therapeutics, Inc., as described in the section entitled “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2025 and subsequent reports to be filed with the SEC. The foregoing sets forth many, but not all, of the factors that could cause actual results to differ from expectations in any forward-looking statement. In light of these risks, uncertainties and assumptions, the forward-looking statements and events discussed in this news release are inherently uncertain and may not occur, and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements. Accordingly, you should not rely upon forward-looking statements as predictions of future events. Except as required by law, we disclaim any intention or responsibility for updating or revising any forward-looking statements. For further information regarding these and other risks related to our business, investors should consult our filings with the SEC, which are available on the SEC's website at www.sec.gov.
All of our pharmaceutical assets under development are investigational product candidates. These have not been approved for use in any medical indication by any regulatory authority in any jurisdiction and their safety, efficacy or other desirable attributes, if any, have not been established in any patient population. Consequently, none of our product candidates is approved or available for sale anywhere in the world.
Our clinical results from earlier-stage clinical trials or preclinical studies may not be indicative of future results from later-stage or larger scale clinical trials and do not ensure regulatory approval. You should not place undue reliance on these statements or any scientific data we present or publish.
We are in the business of new drug discovery and development. Our research and development activities are long, complex, costly and involve a high degree of risk. Holders of our common stock should carefully read our Annual Report on Form 10-K and subsequent Quarterly Reports on Form 10-Q in their entirety, including the risk factors therein. Because risk is fundamental to the process of drug discovery and development, you are cautioned to not invest in our publicly traded securities unless you are prepared to sustain a total loss of the money you have invested.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/877131cb-f6f1-4b81-99c9-d2ea829a9272

Epilepsia paper, Figure 3C
Heatmap of the daily number of seizures per mouse during EEG recordings in each condition. EEG monitoring spanned 20 days, but seizure counts were quantified for the first 5 and last 10 days, and comparative analysis was performed between the first and last 5 days. Gray squares: Mice had died. The 5 day-gap indicates that analysis was not performed for this period.
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.


